The global expansion of pharmaceutical cannabis is reshaping health systems and supply chains worldwide. As regulators, doctors, and patients demand consistent quality, producers face new certification pressures. However, varied pharmacy laws, labeling rules, and export controls still block many market entries. Moreover, centralized EU-GMP manufacturing in hubs such as Lisbon can enable efficient cross-border supply.
In this article we map laboratory-to-patient manufacturing pathways, quality assurance and Qualified Person oversight, in-house cultivation models and cultivation partners, clinical research collaborations, stability and validation timelines, and packaging and labeling best practices; we analyze distribution strategies and finished dosage form approaches for market access, compare regional regulatory frameworks from Germany to France and Eastern Europe, and consider U.S. federal and VA program implications because these elements determine whether pharmaceutical-grade cannabis can reach patients reliably and at scale, and therefore whether companies can build trusted international brands, reduce costs, and accelerate patient access worldwide more quickly.
Global expansion of pharmaceutical cannabis: Market reach
Pharmaceutical cannabis is moving from local pilots to global supply chains. Demand rises because more health systems adopt medical cannabis programs. For example, EU markets such as Germany, the United Kingdom, and Italy now permit widespread distribution. Meanwhile, Australia and New Zealand import EU-GMP products from hubs like Lisbon. As a result, companies can centralize manufacturing and serve many countries with one compliant facility.
Global expansion of pharmaceutical cannabis: Regulatory differences and trade
Regulation still divides markets, however, and this creates barriers. Some countries accept finished dosage forms, while others require magisterial pharmacy compounding. Consequently, labeling, language, and dosage rules differ by market and can delay shipments for months. Manufacturers therefore invest in Quality Assurance and a Qualified Person to meet rules. For broader context on Europe market dynamics, see this analysis here and this follow-up here. Regulators such as the European Medicines Agency play a central role here and the U.S. Food and Drug Administration matters for any federal pathway here.
Global expansion of pharmaceutical cannabis: Emerging trends and opportunities
Several clear trends shape opportunity. First, EU-GMP certification enables cross-border distribution to compliant markets. Second, finished dosage forms are rising because they simplify prescribing and patient use. Third, vertical models mix in-house cultivation with partner farms to secure consistent supply. Moreover, advanced tech like AI seed sorting improves pharma-grade consistency; learn more here. Finally, clinical research partnerships strengthen regulatory submissions and physician trust.
Key takeaways
- Centralized EU-GMP hubs lower production cost and speed export
- Regulatory harmonization would reduce time to market
- Finished dosage forms and rigorous QA build prescriber confidence
These shifts create a pathway for brands to scale globally, because quality, compliance, and trust drive patient access and long term growth.
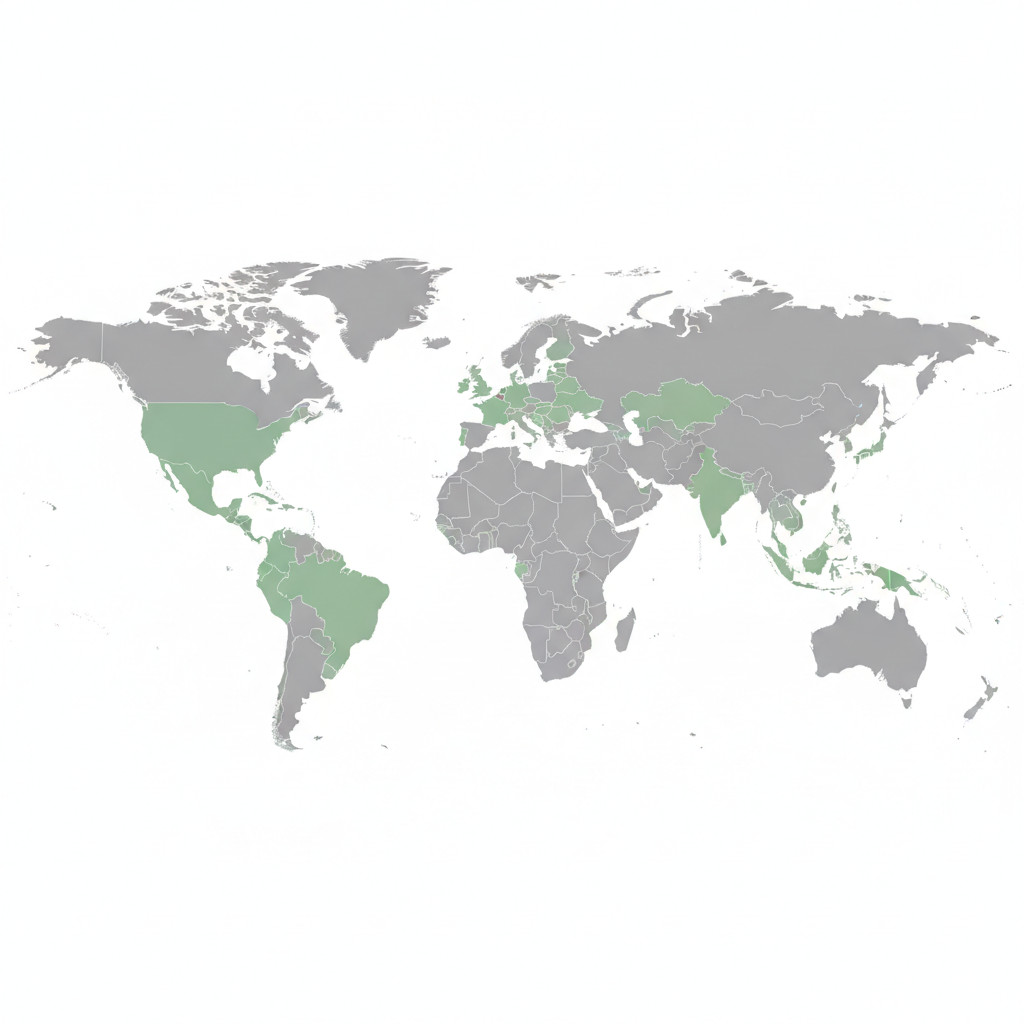
| Country or Region | Regulatory framework | Approval status | Market readiness | Notes and sources |
|---|---|---|---|---|
| European Union region | EU GMP recognized for medicines with national implementation | EU GMP accepted for cross border exports | High | Source: European Medicines Agency guidance. See the EMA site European Medicines Agency |
| Portugal Lisbon hub | National regulator plus EU GMP certified manufacturing sites | Active EU GMP facilities exporting to multiple markets | Very high | Source: Portugal regulator INFARMED. See INFARMED Portugal |
| Germany | National regulator with allowance for magisterial preparations | Controlled; magisterial preparations commonly used | Medium | Source: Federal Ministry of Health overview. See Bundesgesundheitsministerium |
| United Kingdom | MHRA regulation with national prescribing frameworks | Authorized medical cannabis programs | High | Source: Medicines and Healthcare products Regulatory Agency. See MHRA UK |
| France | National regulator with tightly controlled program | Limited authorized products for patient programs | Medium | Source: national health authority documents and program summaries |
| Italy | National regulator with regional implementation variance | Active access in parts of country | Medium variable | Source: regional prescribing and pharmacy rules |
| Australia | TGA oversight for therapeutic goods; imports common | Imports of EU GMP medicines permitted | Medium | Source: Therapeutic Goods regulatory framework and import rules |
| New Zealand | Medsafe regulation with prescriber pathways | Medical cannabis available via prescribers | Medium | Source: Medsafe. See Medsafe NZ |
| Japan | PMDA pharmaceutical pathways and strict approvals | Selective pharmaceutical approvals required | Low to medium | Source: Pharmaceuticals and Medical Devices Agency. See PMDA |
| United States | Federal prohibition with FDA drug approval pathways for registered drugs | Limited federal pathways; active state markets | Variable | Source: U.S. Food and Drug Administration. See FDA |
Mobile friendly stacked cards view
European Union region
EU GMP recognized regionally. Market readiness high. Source EMA link provided above.
Portugal Lisbon hub
EU GMP sites active and export oriented. Readiness very high. See INFARMED link above.
Germany
Permits magisterial preparations; finished dosage rollouts slower. Readiness medium.
United Kingdom
Regulated by MHRA; clinical access evolving. Readiness high.
For other markets refer to the table above and follow the linked national regulator pages for the latest guidance.
Challenges and Opportunities in the global expansion of pharmaceutical cannabis
The pathway to global markets faces heavy regulatory hurdles. Different countries require distinct registrations, therefore timelines can stretch for years. For example, EU-GMP market approvals need about 2.5 years of validation and stability testing before products reach patients. Moreover, labeling and language rules vary, and one error can stop a shipment at customs for months. As a result, companies must invest in Quality Assurance, a Qualified Person, and localized regulatory expertise.
Market access issues also slow growth, however they force strategic innovation. Some nations permit finished dosage forms, while others still require pharmacy compounding. Consequently, manufacturers split strategies between ready-to-dispense medicines and magisterial preparations. In addition, cross-border distribution depends on harmonized standards, so centralized EU-GMP hubs are becoming increasingly valuable for export to Australia, New Zealand, and parts of Latin America.
Societal attitudes create both friction and opportunity. Stigma still surrounds cannabis in some healthcare systems, yet physicians increasingly accept it as a therapeutic option. Therefore, clinical trials and university partnerships are crucial because they build trust with prescribers and regulators. Somai’s Lisbon research collaborations illustrate how clinical evidence can support consistent manufacturing claims and regulatory filings.
Opportunities emerge from these constraints. First, harmonization and finished dosage forms will simplify prescribing and boost patient adherence. Second, vertical supply models combining in-house cultivation with partner farms secure consistent active ingredients. Third, digital and analytical tools such as AI seed sorting and advanced testing raise pharma-grade consistency and reduce batch failures. Finally, firms that master QA, labeling, and regulatory strategy can establish global brands, because trust and compliance ultimately unlock large patient populations and durable market share.
Conclusion
The global expansion of pharmaceutical cannabis shows that quality, compliance, and evidence drive market success. Across regions, EU-GMP hubs and centralized manufacturing lower cost and speed exports. However, national rules and labeling requirements still demand bespoke regulatory strategies. Therefore, firms must pair strong Quality Assurance with clinical research to gain prescriber trust and patient access.
MyCBDAdvisor acts as a trusted, full-spectrum cannabinoid knowledge source, offering news, technical guides, and regulatory insights. Moreover, readers can find practical frameworks and market analysis that clarify complex approval pathways. For example, EMP0 captures emerging market pathways and regulatory milestones. As a result, EMP0 helps readers understand industry evolution and prioritize next steps for market entry.
If you explore pharmaceutical cannabis, start with evidence-based planning. Use clinical partnerships, invest in EU-GMP where relevant, and build clear labeling and distribution plans. Finally, visit MyCBDAdvisor for ongoing guidance and resources. We encourage professionals and stakeholders to stay informed, because informed action accelerates patient access and global growth.
Frequently Asked Questions (FAQs)
What does pharmaceutical cannabis mean and how does it differ from other cannabis products?
Pharmaceutical cannabis refers to medicines made to pharma standards. Manufacturers follow strict manufacturing and testing rules. As a result, products have consistent potency and safety profiles. Therefore, doctors can prescribe them with more confidence.
Why do regulations vary so much between countries and what should exporters expect?
Regulatory systems reflect different legal and medical frameworks. For example, some countries allow finished dosage forms, while others require pharmacy compounding. Consequently, exporters must tailor labeling, language, and documentation for each market. Expect longer timelines and additional local approvals.
What is EU-GMP and why is it important for global expansion?
EU-GMP stands for Good Manufacturing Practice in the EU. It proves that a facility meets pharmaceutical quality standards. Because of this, EU-GMP certification enables central manufacturing to supply multiple compliant markets. In short, it reduces duplicates and speeds cross-border exports.
How long does it take for a pharmaceutical cannabis product to reach patients?
Timelines vary by market and dossier complexity. However, EU-style market approvals often need around two and a half years for validation and stability testing. Clinical studies and local registration steps can add time. Planning and parallel workstreams reduce delays.
What practical steps can companies take to improve market access and patient trust?
Start with robust Quality Assurance and a Qualified Person oversight. Build clinical partnerships to generate evidence because evidence strengthens regulatory filings. Invest in labeling accuracy and local language leaflets. Finally, choose distribution partners who understand doctors and pharmacies.
If you have more questions, consult MyCBDAdvisor for technical guides and regulatory summaries.
















